
Tramadol cas No. 27203-92-5
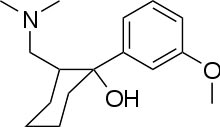 Systematic
(IUPAC) name Systematic
(IUPAC) name
(1R,2R)-rel-2-[(dimethylamino)methyl]-
1-(3-methoxyphenyl)cyclohexanol
Identifiers
CAS number : 27203-92-5
ATC code : N02AX02
PubChem : 33741
DrugBank : APRD00028
ChemSpider : 31105
Chemical data
Formula : C16H25NO2
Mol. mass : 263.4 g/mol
SMILES : eMolecules & PubChem
Pharmacokinetic data
Bioavailability 68–72% Increases with repeated dosing.
Protein binding 20%
Metabolism Hepatic demethylation and glucuronidation
Half life 5–7 hours
Excretion Renal
Tramadol is a centrally acting analgesic, used for treating
moderate to severe pain. It is often categorized as an opioid, although
it is chemically not related to the opioid class of drugs. It does,
however, appear to have agonist actions at the μ-opioid receptor as well
as the noradrenergic and serotonergic systems.
Tramadol is used to treat moderate to moderately severe pain and most
types of neuralgia, including trigeminal neuralgiaIt has been suggested
that tramadol could be effective for alleviating symptoms of depression,
anxiety, and phobias because of its action on the noradrenergic and
serotonergic systems.However, health professionals have not yet fully
endorsed of its use on a large scale for these disorders, although it
may be used when other treatments have failed.
 Tramadol
is a centrally acting analgesic. The chemical name for tramadol
hydrochloride is
(±)cis-2-[(dimethylamino)methyl]-1-(3-methoxyphenyr) cyclohexanol
hydrochloride.Tramadol hydrochloride is a white, bitter, crystalline and
odorless powder. It is readily soluble in water and ethanol and has a
pKa of 9.41. The n-octanol/water log partition coefficient (logP) is
1.35 at pH 7. Tramadol
is a centrally acting analgesic. The chemical name for tramadol
hydrochloride is
(±)cis-2-[(dimethylamino)methyl]-1-(3-methoxyphenyr) cyclohexanol
hydrochloride.Tramadol hydrochloride is a white, bitter, crystalline and
odorless powder. It is readily soluble in water and ethanol and has a
pKa of 9.41. The n-octanol/water log partition coefficient (logP) is
1.35 at pH 7.
How should this medicine be used?
Tramadol comes as a tablet and an extended-release (long-acting) tablet
to take by mouth. The regular tablet is usually taken with or without
food every 4-6 hours as needed. The extended-release tablet should be
taken once a day. Take the extended-release tablet at about the same
time of day every day, and either always take it with food or always
take it without food. Take tramadol exactly as directed. Do not take
more medication as a single dose or take more doses per day than
prescribed by your doctor. Taking more tramadol than prescribed by your
doctor may cause serious side effects or death.
Your doctor may start you on a low dose of tramadol and gradually
increase the amount of medication you take, not more often than every 3
days if you are taking the regular tablets or every 5 days if you are
taking the extended-release tablets.
Swallow the extended-release tablets whole; do not split, chew, or crush
them. Do not snort (inhale powder from crushed tablet) or inject the
dissolved extended-release tablets. Taking this medication in a way that
is not recommended may cause serious side effects or death.
Tramadol can be habit-forming. Do not take a larger dose, take it more
often, or take it for a longer period of time than prescribed by your
doctor. Call your doctor if you find that you want to take extra
medication or if you notice any other unusual changes in your behavior
or mood.
Tramadol side effects
Get emergency medical help if you have any of these signs of an allergic
reaction: hives; difficulty breathing; swelling of your face, lips,
tongue, or throat. Stop using tramadol and call your doctor at once if
you have any of these serious side effects:
* seizure (convulsions);
* a red, blistering, peeling skin rash; or
* shallow breathing, weak pulse.
Less serious side effects may include:
* dizziness, drowsiness, weakness;
* nausea, vomiting, constipation, loss of appetite;
* blurred vision;
* flushing (redness, warmth, or tingly feeling); or
* sleep problems (insomnia).
Availability
Tramadol is usually marketed as the hydrochloride salt (tramadol
hydrochloride); the tartrate is seen on rare occasions, and tramadol is
available in both injectable (intravenous and/or intramuscular) and oral
preparations. It is also available in conjunction with acetaminophen.
The solutions suitable for injection are used in Patient-Controlled
Analgesia pumps under some circumstances, either as the sole agent or
along with another agent such as morphine.
Tramadol comes in many forms, including:
* capsules
* tablets
* extended-release tablets
* extended-release capsules
* chewable tablets
* low-residue and/or uncoated tablets that can be taken by the
sublingual and buccal routes
* suppositories
* effervescent tablets and powders
* ampoules of sterile solution for SC, IM, and IV injection
* preservative-free solutions for injection by the various spinal routes
(epidural, intrathecal, caudal, and others)
* powders for compounding
* liquids both with and without alcohol for oral and sublingual
administration, available in regular phials and bottles, dropper
bottles, bottles with a pump similar to those used with liquid soap and
phials with droppers built into the cap
* tablets and capsules containing paracetamol (acetaminophen) and
aspirin and other agents
Tramadol has been experimentally used in the form of an ingredient in
multi-agent topical gels, creams, and solutions for nerve pain, rectal
foam, concentrated retention enaema, and a skin plaster (transdermal
patch) quite similar to those used with lidocaine.
Veterinary Use
Tramadol is used to treat post-operative, injury-related, and chronic
(e.g., cancer-related) pain in dogs and cats [8] as well as rabbits,
coatis, many small mammals including rats and flying squirrels, guinea
pigs, ferrets, and raccoons. Tramadol comes in ampoules in addition to
the tablets, capsules, powder for reconstitution, and oral syrups and
liquids; the fact that its characteristic taste is not very bitter and
can be masked in food and diluted in water makes for a number of means
of administration. No data that would lead to a definitive determination
of the efficacy and safety of tramadol in reptiles or amphibians is
available at this time, and, following the pattern of all other drugs,
it appears that tramadol can be used to relieve pain in marsupials such
as North American opossums, Short-Tailed Opossums, sugar gliders,
wallabies, and kangaroos among others.
Metabolism
Tramadol undergoes hepatic metabolism via the cytochrome P450 isozyme
CYP2D6, being O- and N-demethylated to five different metabolites. Of
these, M1 (O-Desmethyltramadol) is the most significant since it has 200
times the μ-affinity of (+)-tramadol, and furthermore has an elimination
half-life of nine hours, compared with six hours for tramadol itself. In
the 6% of the population that have slow CYP2D6 activity, there is
therefore a slightly reduced analgesic effect. Phase II hepatic
metabolism renders the metabolites water-soluble, which are excreted by
the kidneys. Thus, reduced doses may be used in renal and hepatic
impairment.

 Note:
These API/ chemicals are designated as
those that are used in the manufacture of the controlled substances and
are important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your country
government laws /control substance ACT. Note:
These API/ chemicals are designated as
those that are used in the manufacture of the controlled substances and
are important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your country
government laws /control substance ACT.
Note /Government Notification:
These chemicals are designated as those that are used in the manufacture
of the controlled substances and are important to the manufacture of the
substances. For any (Control Substance) products Import and Export ***
subjected to your country government laws /control substance ACT.
Information: The information on this web page is provided to help you
to work safely, but it is intended to be an overview of hazards, not a
replacement for a full Material Safety Data Sheet (MSDS). MSDS forms can
be downloaded from the web sites of many chemical suppliers. ,also that
the information on the PTCL Safety web site, where this page was hosted,
has been copied onto many other sites, often without permission. If you
have any doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web browser
displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in Physical
Chemistry at Oxford University. If not, this page is a copy made by some
other person and we have no responsibility for it.
The Controlled Substances Act (CSA) was enacted into law by the Congress
of the United States as Title II of the Comprehensive Drug Abuse
Prevention and Control Act of 1970.[1] The CSA is the federal U.S. drug
policy under which the manufacture, importation, possession, use and
distribution of certain substances is regulated. The Act also served as
the national implementing legislation for the Single Convention on
Narcotic Drugs.
|
|




 Systematic
(IUPAC) name
Systematic
(IUPAC) name  Tramadol
is a centrally acting analgesic. The chemical name for tramadol
hydrochloride is
(±)cis-2-[(dimethylamino)methyl]-1-(3-methoxyphenyr) cyclohexanol
hydrochloride.Tramadol hydrochloride is a white, bitter, crystalline and
odorless powder. It is readily soluble in water and ethanol and has a
pKa of 9.41. The n-octanol/water log partition coefficient (logP) is
1.35 at pH 7.
Tramadol
is a centrally acting analgesic. The chemical name for tramadol
hydrochloride is
(±)cis-2-[(dimethylamino)methyl]-1-(3-methoxyphenyr) cyclohexanol
hydrochloride.Tramadol hydrochloride is a white, bitter, crystalline and
odorless powder. It is readily soluble in water and ethanol and has a
pKa of 9.41. The n-octanol/water log partition coefficient (logP) is
1.35 at pH 7.




